Optimisation of Chondrocyte Propagation from Human Articular Cartilage to Preserve the Chondrocyte characteristics
Main Article Content
Abstract
Introduction: Osteoarthritis (OA) is a major health burden and affects millions of people worldwide. This occurs when the cartilage degenerates over a period of time and OA commonly affects the joints in the hand, hips, and knees. Autologous chondrocyte implantation (ACI) and Matrix, matrix-associated autologous chondrocyte implantation (MACI) is a procedure used to treat isolated full-thickness articular cartilage defects of the knee. The cornerstone of these procedures is the ability to cultivate and grow a large number of chondrocytes for a successful outcome.
Materials and Methods: Cartilage specimens of patients undergoing total knee arthroplasty (TKA) were digested using standard collagenase treatment. The resulting cells isolated were evaluated for viability. The cells at equal seeding density were plated on different media for evaluation. Cells from each passage were counted, assessed for viability, immunophenotyping utilizing surface markers used for the characterization of chondrocytes and RT-PCR was performed to confirm expression of chondrocyte genes.
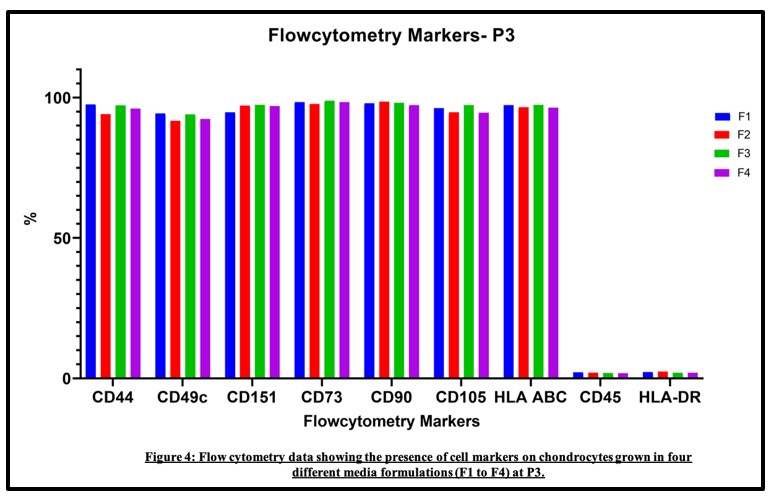
Results: Primary cells plated in DMEM F12 attained faster confluency, the addition of ITS supplement was not essential. Cells maintained their chondrocyte morphology and surface marker expression over passages and expressed genes for chondrocytes. Additional 5 samples were cultured on DMEM F12 without ITS and found to maintain chondrocyte characteristics.
Clinical Significance: Chondrocytes are crucial not only for the development of therapeutic approaches in cartilage repair but are necessary in cartilage tissue engineering to allow the development of functional cell models and novel scaffolds. As a result, an optimized, proven method for chondrocyte isolation must be developed and shared with other groups.
Conclusion: In this study, we describe a simple and affordable procedure of isolation and cultivation of human articular chondrocytes that demonstrated a high chondrogenic potential to the third passage, which is sufficient to grow cells for an ACI procedure.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
The Journals licensing terms of CC-BY-NC-SA 4.0 speaks that you are free to Share (copy and redistribute the material in any medium or format), Adapt (remix, transform, and build upon the material) under proper terms of Attribution (You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.), NonCommercial (You may not use the material for commercial purposes.), ShareAlike (If you remix, transform, or build upon the material, you must distribute your contributions under the same license as the original.) and No additional restrictions (You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.)
References
Vedicherla S, Buckley CT. Rapid chondrocyte isolation for tissue engineering applications: The effect of enzyme concentration and temporal exposure on the matrix forming capacity of nasal derived chondrocytes. Biomed Res Int [Internet]. 2017;2017:2395138. Available from: http://dx.doi.org/10.1155/2017/2395138
Kon E, Filardo G, Di Martino A, Marcacci M. ACI and MACI. J Knee Surg [Internet]. 2012;25(01):017–22. Available from: http://dx.doi.org/10.1055/s-0031-1299651
Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med [Internet]. 1994;331(14):889–95. Available from: http://dx.doi.org/10.1056/NEJM199410063311401
Brittberg M, Sjögren-Jansson E, Thornemo M, Faber B, Tarkowski A, Peterson L, et al. Clonal growth of human articular cartilage and the functional role of the periosteum in chondrogenesis. Osteoarthritis Cartilage [Internet]. 2005;13(2):146–53. Available from: http://dx.doi.org/10.1016/j.joca.2004.10.020
Tallheden T, Bengtsson C, Brantsing C, Sjögren-Jansson E, Carlsson L, Peterson L, et al. Proliferation and differentiation potential of chondrocytes from osteoarthritic patients. Arthritis Res Ther [Internet]. 2005;7(3):R560-8. Available from: http://dx.doi.org/10.1186/ar1709
Oseni AO, Butler PE, Seifalian AM. Optimization of chondrocyte isolation and characterization for large-scale cartilage tissue engineering. J Surg Res [Internet]. 2013;181(1):41–8. Available from: http://dx.doi.org/10.1016/j.jss.2012.05.087
Jakob M, Démarteau O, Schäfer D, Stumm M, Heberer M, Martin I. Enzymatic digestion of adult human articular cartilage yields a small fraction of the total available cells. Connect Tissue Res [Internet]. 2003;44(3–4):173–80. Available from: http://dx.doi.org/10.1080/03008200390215836
Schulze-Tanzil G, de Souza P, Villegas Castrejon H, John T, Merker H-J, Scheid A, et al. Redifferentiation of dedifferentiated human chondrocytes in high-density cultures. Cell Tissue Res [Internet]. 2002;308(3):371–9. Available from: http://dx.doi.org/10.1007/s00441-002-0562-7
Stall NM, Kagoma YK, Bondy JN, Naudie D. Surgical waste audit of 5 total knee arthroplasties. Can J Surg [Internet]. 2013;56(2):97–102. Available from: http://dx.doi.org/10.1503/cjs.015711
Schmid I, Krall WJ, Uittenbogaart CH, Braun J, Giorgi JV. Dead cell discrimination with 7-amino-actinomycin D in combination with dual color immunofluorescence in single laser flow cytometry. Cytometry [Internet]. 1992;13(2):204–8. Available from: http://dx.doi.org/10.1002/cyto.990130216
Grogan SP, Barbero A, Diaz-Romero J, Cleton-Jansen A-M, Soeder S, Whiteside R, et al. Identification of markers to characterize and sort human articular chondrocytes with enhanced in vitro chondrogenic capacity. Arthritis Rheum [Internet]. 2007;56(2):586–95. Available from: http://dx.doi.org/10.1002/art.22408
Diaz-Romero J, Gaillard JP, Grogan SP, Nesic D, Trub T, Mainil-Varlet P. Immunophenotypic analysis of human articular chondrocytes: changes in surface markers associated with cell expansion in monolayer culture. J Cell Physiol [Internet]. 2005;202(3):731–42. Available from: http://dx.doi.org/10.1002/jcp.20164
Tendon-Derived ProgenitorCells With Multilineage Potential Are Present Within Human Patellar Tendon. The Orthopaedic Journal of Sports Medicine. 2021(8).
Oeyen AL, Kircher J, Vogl M, Ickert I, Osada N, Krauspe R, et al. Dexamethasone does not compensate for local anesthetic cytotoxic effects on tenocytes: Morphine or morphine plus dexamethasone may be a safe alternative. Arthrosc Sports Med Rehabil [Internet]. 2022;4(2):e459–69. Available from: http://dx.doi.org/10.1016/j.asmr.2021.11.004
Schnabel M, Marlovits S, Eckhoff G, Fichtel I, Gotzen L, Vécsei V, et al. Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage [Internet]. 2002;10(1):62–70. Available from: http://dx.doi.org/10.1053/joca.2001.0482
Hayman DM, Blumberg TJ, Scott CC, Athanasiou KA. The effects of isolation on chondrocyte gene expression. Tissue Eng [Internet]. 2006;12(9):2573–81. Available from: http://dx.doi.org/10.1089/ten.2006.12.2573
Hu D-N, Yang P-Y, Ku M-C, Chu C-H, Lim AY, Hwang M-H. Isolation and cultivation of human articular chondrocytes. Kaohsiung J Med Sci. 2002;18(3):113–20.
Laganà M, Arrigoni C, Lopa S, Sansone V, Zagra L, Moretti M, et al. Characterization of articular chondrocytes isolated from 211 osteoarthritic patients. Cell Tissue Bank [Internet]. 2014;15(1):59–66. Available from: http://dx.doi.org/10.1007/s10561-013-9371-3
Strzelczyk P, Benke G, Górecki A. Methods for the isolation and culture of human articular chondrocytes. Ortop Traumatol Rehabil. 2001;3(2):213–5.
Amrichová J, Špaková T, Rosocha J, Harvanová D, Bacenková D, Lacko M. Effect of PRP and PPP on proliferation and migration of human chondrocytes and synoviocytes in vitro. Central European Journal of Biology. 2014;9(2):139–48.
Wang F, Zhang B, Yang H, Li A, Li R, Zhou D. Transforming growth factor-beta can induce differentiation of hypertrophic chondrocytes into osteoblast-like cells in epiphyseal plate. Hua Xi Yi Ke Da Xue Xue Bao. 2001;32(2):243–6.
Summers KL, O’Donnell JL, Hoy MS, Peart M, Dekker J, Rothwell A, et al. Monocyte-macrophage antigen expression on chondrocytes. J Rheumatol. 1995;22(7):1326–34.